Z. Li, K. Mathwig, O. A. Arotiba, L. Tshwenya , E. B. Carneiro-Neto, E. C. Pereira, F. Marken, Curr. Opin. Electrochem. 39 (2023) 101280. [link, pdf]
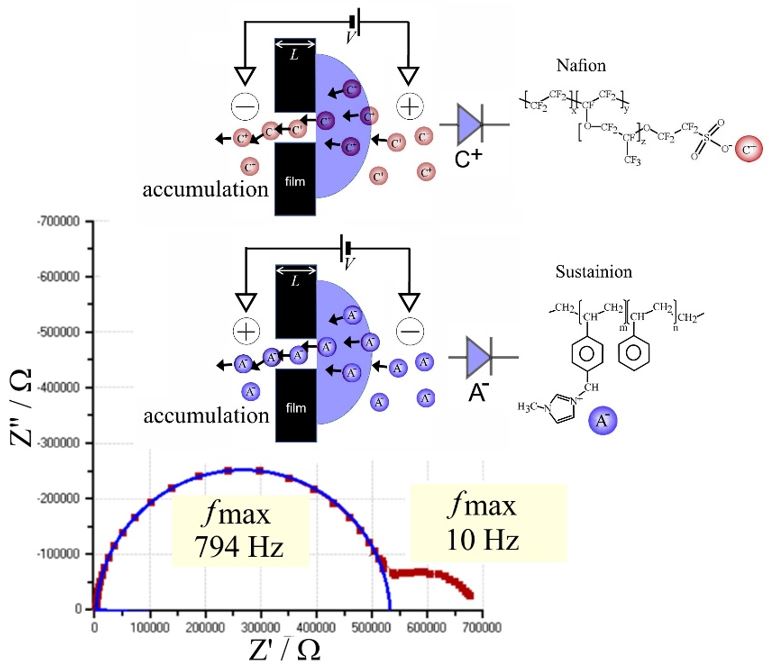
Ionic diodes have emerged repeatedly in the literature for gel interfaces, for nanopores and channels, for nano-/micro-fluidic systems, and for asymmetrically ionomer-covered microholes. Concentration polarisation is likely to be the key to understanding the diode function and the diode time constant τdiode, i.e. the time for approaching steady state following a potential/polarity switch. For frequencies higher than ωdiode = 2πf = (τdiode)-1, the polarization mechanism is too slow for ion current rectification. Below the frequency associated with the diode time constant, irreversibility in ion flow is induced and the diode switches between two resistive states at opposite potentials (“open” and “closed”). The irreversible flow of ions allows energy conversion from electrical to electrochemical. For energy conversion, two coupled ionic diodes are necessary driven by alternating current (AC) electricity to minimise driver electrode electrolysis and energy losses. Opportunities for AC-desalination and for electroosmotic water harvesting with coupled ionic diodes are discussed.