Z. Li, T. Pang. J. Shen, P. J. Fletcher, K. Mathwig, F. Marken, Micro and Nano Engineering 16 (2022) 100157. [link, pdf]
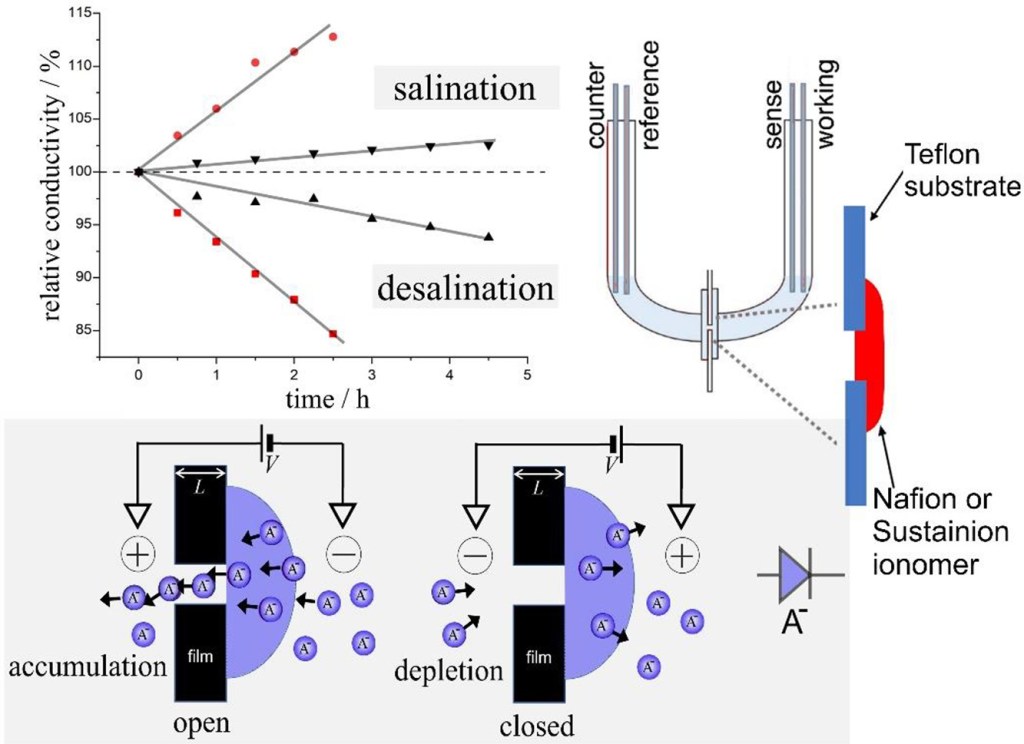
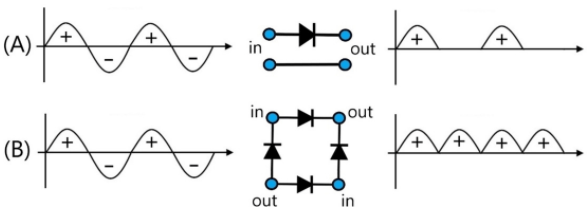
Microscale ionic rectifier effects are commonly observed in devices based on semipermeable ionomer coated on an array of microholes with potential applications in alternating current (AC) driven desalination and/or electroosmotic pumping. The efficiency of devices is dependent on ionic diode switching speed, the rectification ratio, and the design of materials and the ionic circuit. Here, a new circuit is proposed based on coupling in parallel (i) a cationic diode based on the cation conductor Nafion and (ii) an anionic diode based on the anion conducting Sustainion. With an alternating driving voltage, a net desalination effect is observed without any moving parts and without significant side reactions. Experimentally, a 4-electrode configuration and a 2-electrode configuration are compared. The ionic diode desalination system is shown to work with only two carbon mat driver electrodes, but the performance in particular at higher ionic strengths (>10 mM) still needs to be improved. Based on the experimental prototype, the current/power efficiency are investigated and challenges for future improvements are discussed.


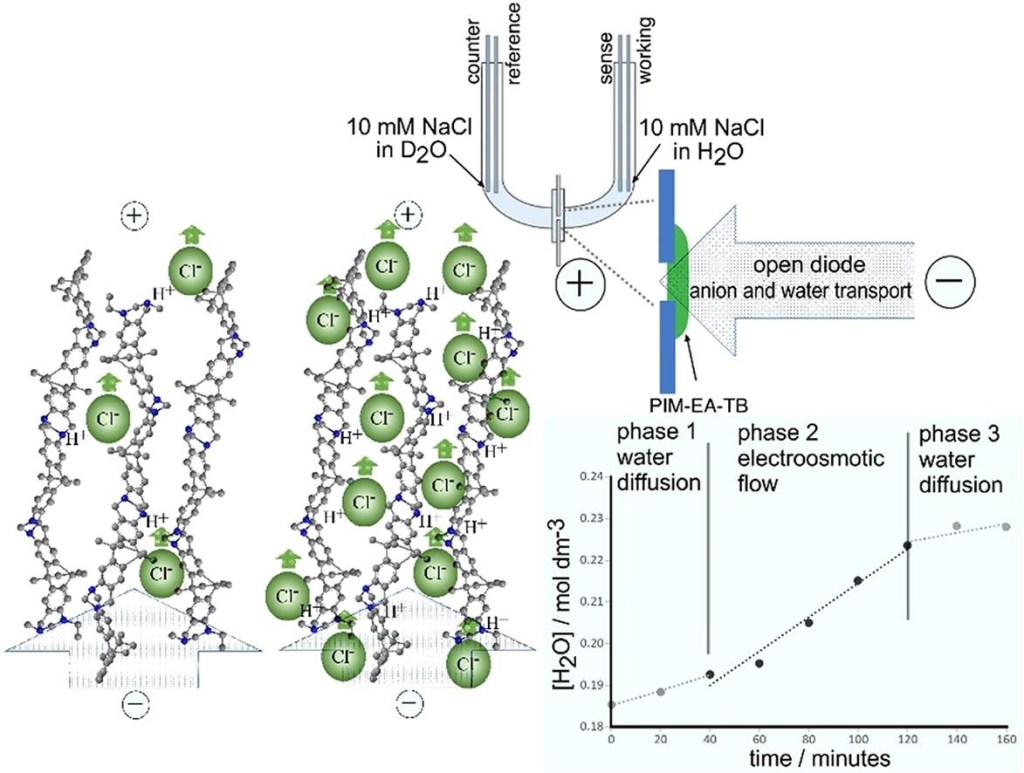
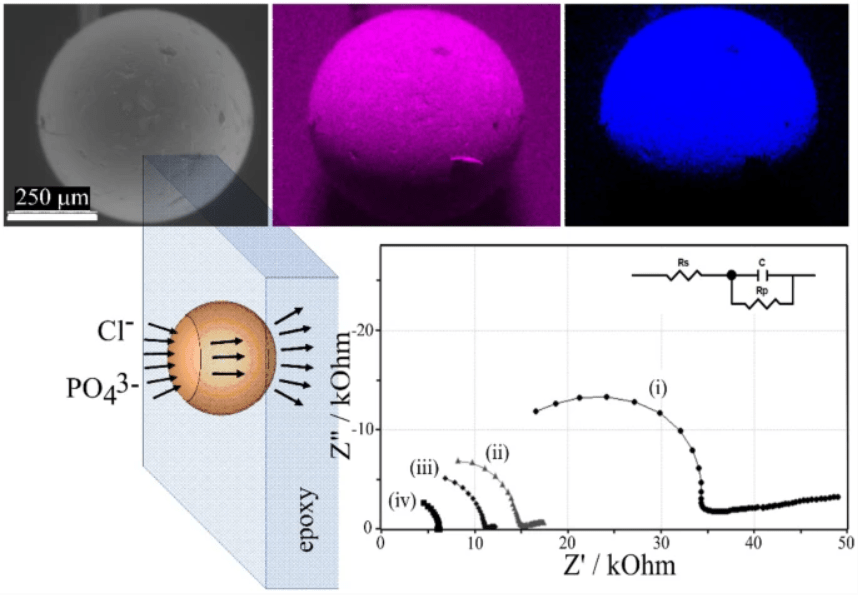
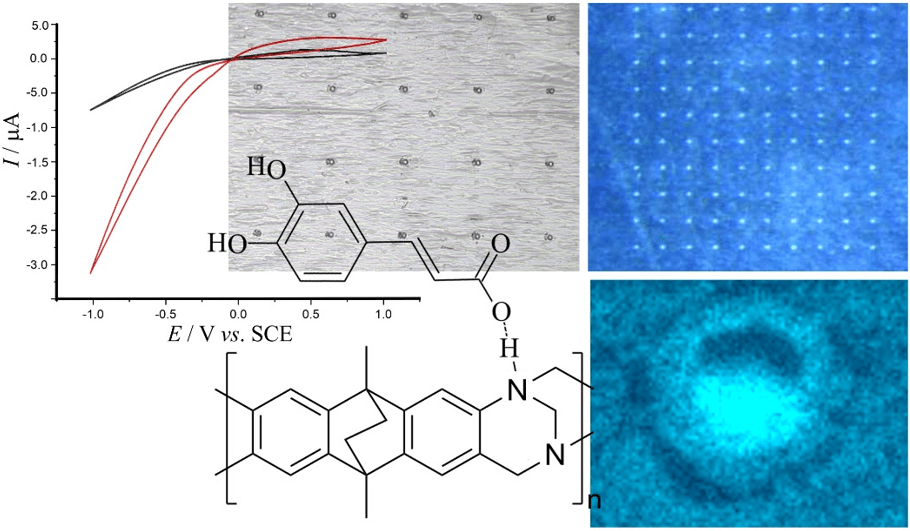

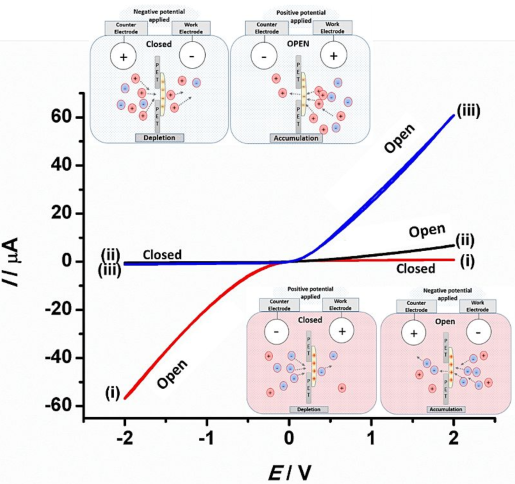 Membrane materials with semi-permeability for anions or for cations are of interest in electrochemical and nanofluidic separation and purification technologies. In this study, partially hydrolyzed poly-acrylonitrile (phPAN) is investigated as a pH-switchable anion/cation conductor. When switching from anionic to cationic semi-permeability, also the ionic current rectification effect switches for phPAN materials deposited asymmetrically onto a 5, 10, 20, or 40 µm diameter microhole in a 6 µm thick polyethylene-terephthalate (PET) film substrate. Therefore, ionic rectifier behavior can be tuned and used to monitor and characterize semi-permeability. Effects of electrolyte type and concentration, and pH (relative to the zeta potential at approximately 3.1) are investigated by voltammetry, chronoamperometry, and impedance spectroscopy. A computational model provides good qualitative agreement with observed electrolyte concentration data. High rectification effects are observed for both cations (pH > 3.1) and anions (pH < 3.1), but only at relatively low ionic strengths.
Membrane materials with semi-permeability for anions or for cations are of interest in electrochemical and nanofluidic separation and purification technologies. In this study, partially hydrolyzed poly-acrylonitrile (phPAN) is investigated as a pH-switchable anion/cation conductor. When switching from anionic to cationic semi-permeability, also the ionic current rectification effect switches for phPAN materials deposited asymmetrically onto a 5, 10, 20, or 40 µm diameter microhole in a 6 µm thick polyethylene-terephthalate (PET) film substrate. Therefore, ionic rectifier behavior can be tuned and used to monitor and characterize semi-permeability. Effects of electrolyte type and concentration, and pH (relative to the zeta potential at approximately 3.1) are investigated by voltammetry, chronoamperometry, and impedance spectroscopy. A computational model provides good qualitative agreement with observed electrolyte concentration data. High rectification effects are observed for both cations (pH > 3.1) and anions (pH < 3.1), but only at relatively low ionic strengths. In ion-annihilation electrochemiluminescence (ECL), luminophore ions are generated by oxidation as well as reduction at electrodes surfaces, and subsequently recombine into an electronically excited state, which emits light. The intensity of the emitted light is often limited by the kinetic rate of recombination of the luminophore ion species. Recombination or annihilation rates are high ranging up to approximately 1010 M−1 s−1 and can be difficult to determine using scanning electrochemical microscopy or high-frequency oscillations of an electrode potential. Here, we propose determining annihilation kinetics by measuring the relative change of the emitted light intensity as a function of luminophore concentration. Using finite element simulations of annihilation ECL in a geometry of two closely spaced electrodes biased at constant potentials, we show that, with increasing concentrations, luminescence intensity crosses over from a quadratic dependence on concentration to a linear regime—depending on the rate of annihilation. Our numerical results are applicable to scanning electrochemical microscopy as well as nanofluidic electrochemical devices to determine fast ion-annihilation kinetics.
In ion-annihilation electrochemiluminescence (ECL), luminophore ions are generated by oxidation as well as reduction at electrodes surfaces, and subsequently recombine into an electronically excited state, which emits light. The intensity of the emitted light is often limited by the kinetic rate of recombination of the luminophore ion species. Recombination or annihilation rates are high ranging up to approximately 1010 M−1 s−1 and can be difficult to determine using scanning electrochemical microscopy or high-frequency oscillations of an electrode potential. Here, we propose determining annihilation kinetics by measuring the relative change of the emitted light intensity as a function of luminophore concentration. Using finite element simulations of annihilation ECL in a geometry of two closely spaced electrodes biased at constant potentials, we show that, with increasing concentrations, luminescence intensity crosses over from a quadratic dependence on concentration to a linear regime—depending on the rate of annihilation. Our numerical results are applicable to scanning electrochemical microscopy as well as nanofluidic electrochemical devices to determine fast ion-annihilation kinetics.